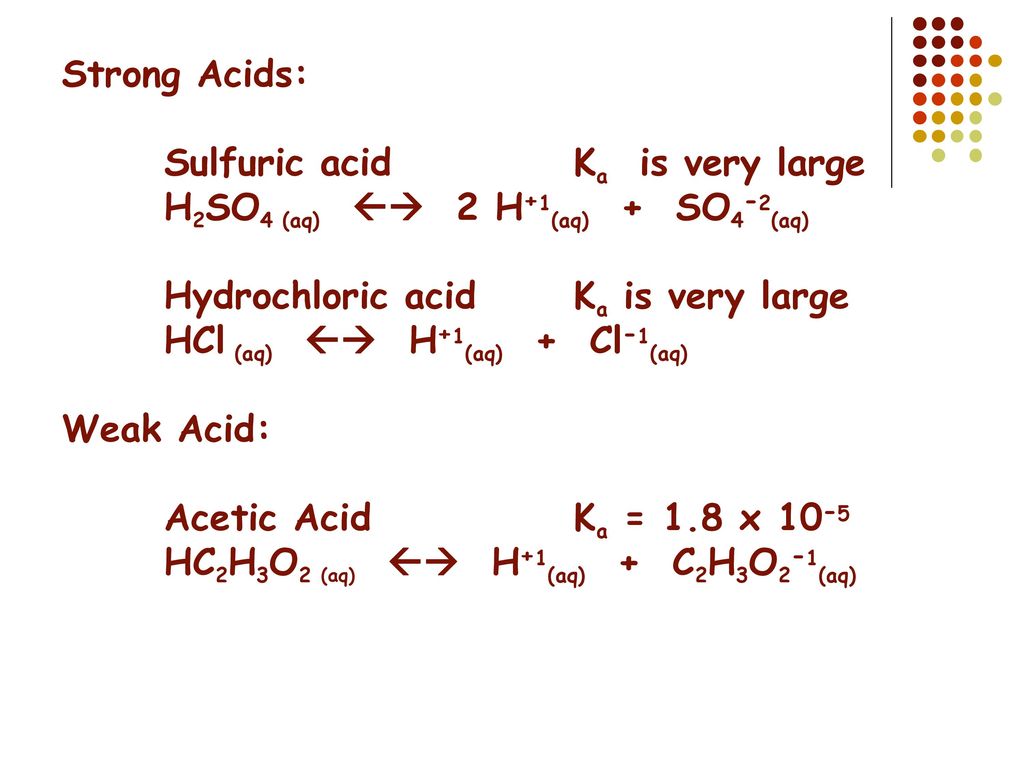
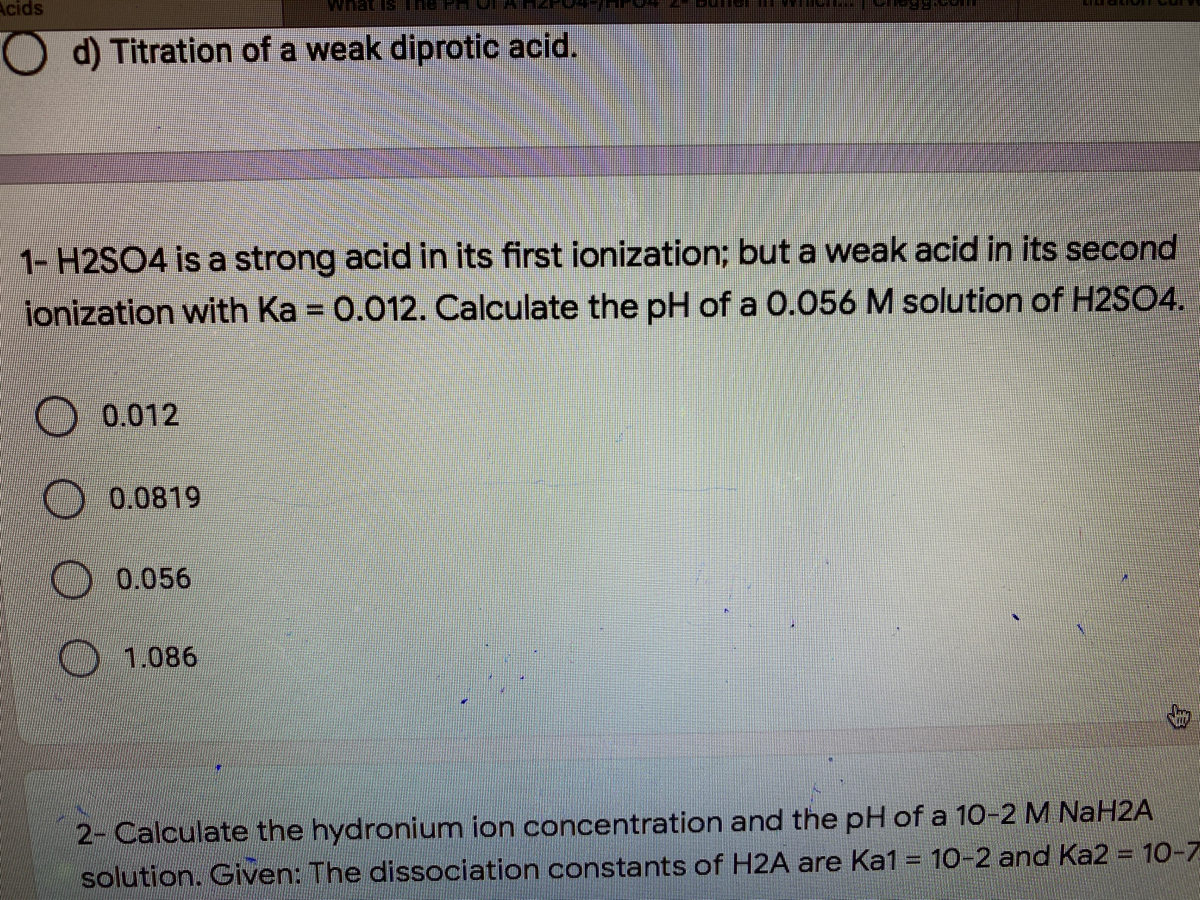
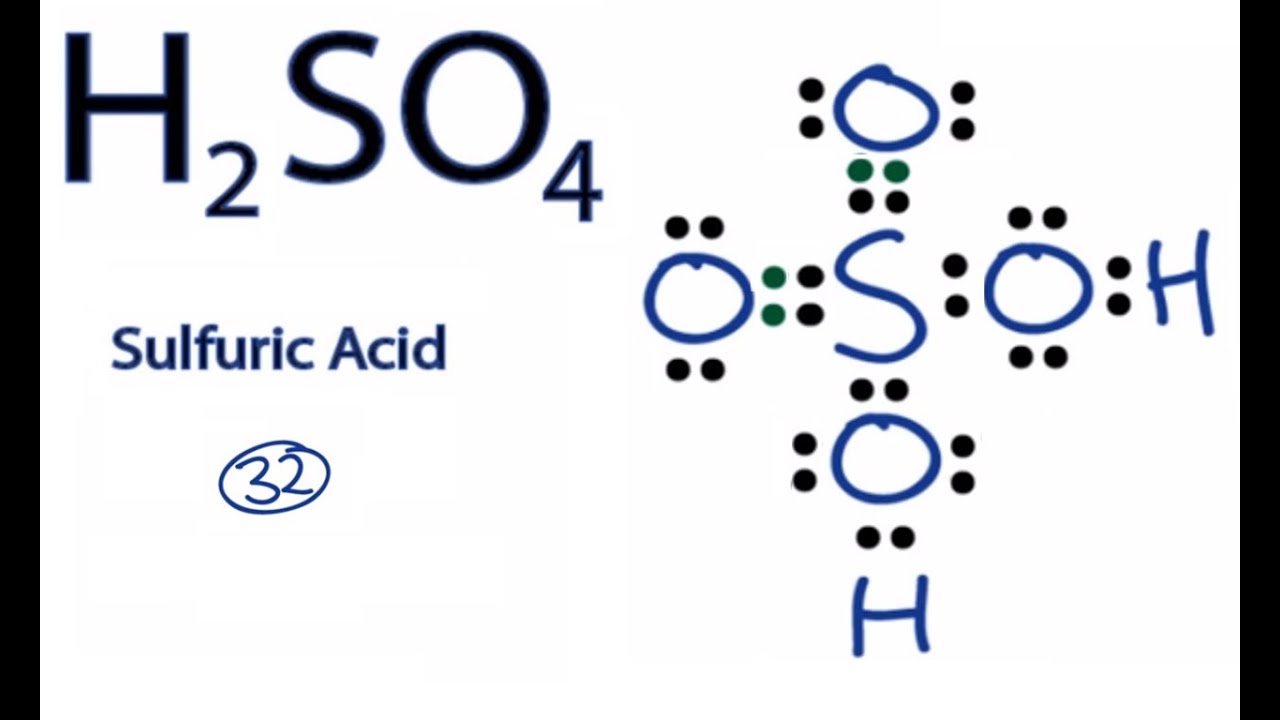
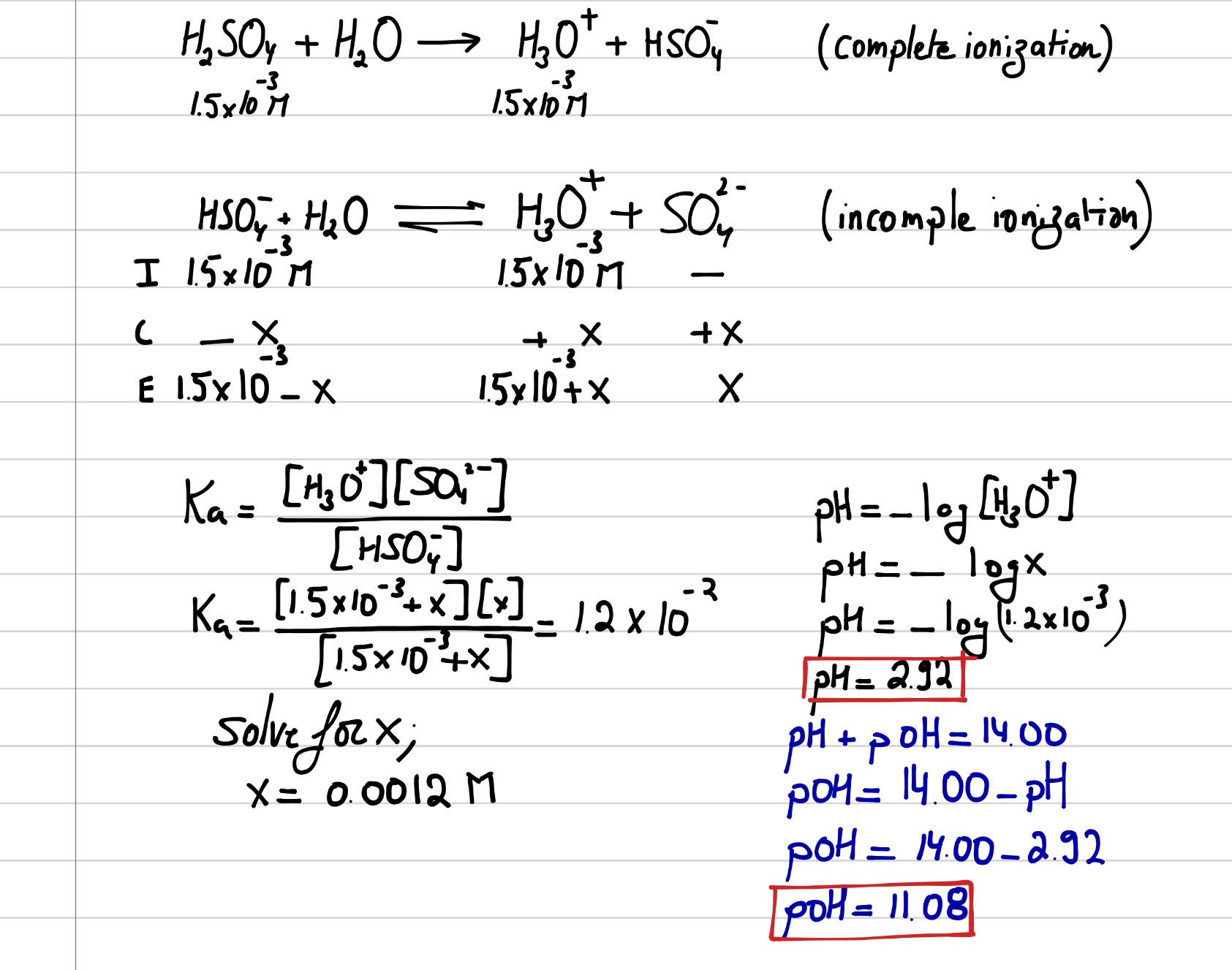SOLVED: PH of Aqueous Sulfuric Acid Calculate the pH of a 3.12x10^-3 M solution of H2SO4: (Ka = 0.0120 for HSO4)

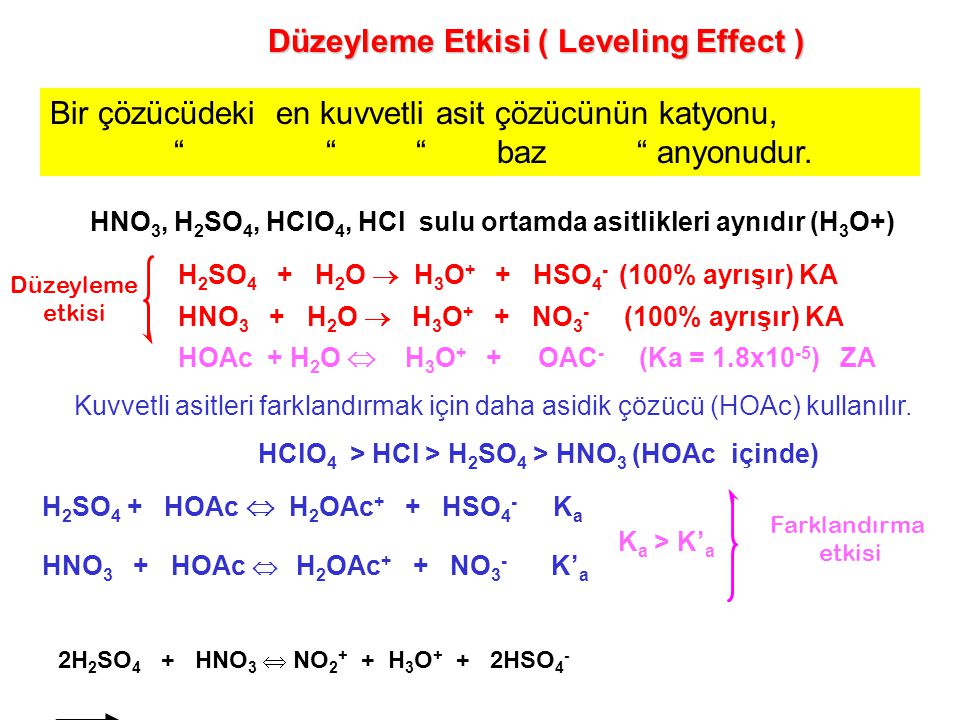
Poliprotik Asitler Bünyesinde birden fazla iyonlaşabilen hidrojen içeren asitlerdir. Örneğin H2SO4,H3PO4 ve H2CO3 gibi … H3A şeklindeki bir poliprotik. - ppt indir